Unmatched Robust, Invisible Activity Recording
Operating invisibly, record EVERYTHING your child or your employee does with SpyAgent's wide-array of 50+ computer monitoring features.
Operating invisibly, record EVERYTHING your child or your employee does with SpyAgent's wide-array of 50+ computer monitoring features.
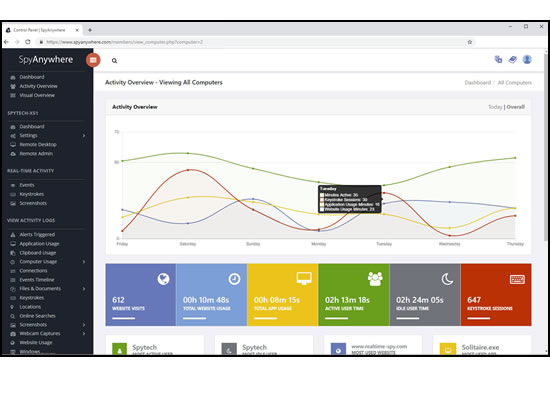
View activities in real-time from anywhere via your browser. Receive email reports and real-time alerts. Remotely uninstall from the cloud!
SpyAgent turns 25 in 2025 which means we have had lots of time and feedback to make an extremely refined computer monitoring solution.
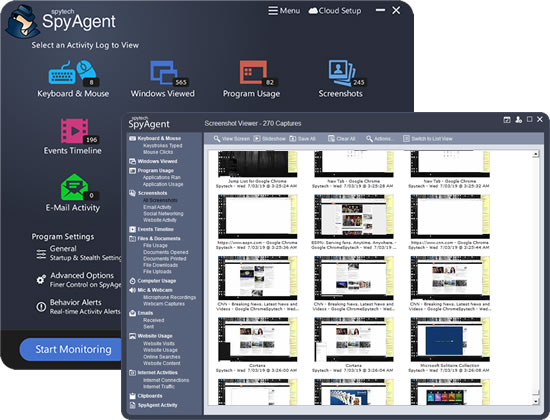
SpyAgent's unmatched all-seeing eye can bring an array of benefits to your family or business environment. With the ability to log all keystrokes, track web and program usage down to the second, and show you everything that has happened with screenshots, SpyAgent helps you learn the truth and put your mind at ease!
SpyAgent's main purpose is to record everything your child or employee does. Here's what it records.
SpyAgent's keylogger logs everything users type - including passwords.
Log what apps are ran, and for how long they are actually interacted with.
Log all visits and online searches, and see how long each page was visited.
Visual logging of everything done, played back in a convenient slideshow.
Record what is happening around your computer, as well as on it.
Capture images from the webcam to see who is using your computer.
See all social network activity, email messages, and chat sessions.
Track how long your computer is used, and how long users are active.
A chronological timeline of everything that has happened on your computer.
Log internet connections established, and even actual raw internet traffic data.
Log what files are used, copied, renamed, deleted, and even transferred.
Log every mouse click action, along with where it was clicked.
SpyAgent is not just a full-featured computer monitoring solution; it's feature set goes above and beyond just monitoring and includes many more useful features - like comprehensive activity filtering, real-time behavior alerts, cloud access, smart logging, self-destruct uninstall, graphical log reports, and more!
SpyAgent can block websites, chat clients, and applications used. It can alert you in real-time when filters are triggered, and when keywords are typed.
Activity triggered monitoring and screenshot captures provide flexible logging. SpyAgent's report generator provides useful Top-10 and 'Most Popular' reports.
SpyAgent provides powerful built-in log viewers for local access and management, as well as cloud access and log deliveries via email and FTP for remote monitoring.
Besides being the most full-featured computer monitoring solution available, here are some more reasons to choose SpyAgent.

Top10Reviews.com

T5A.com

Keylogger.org
SpyAgent is developed and supported by Spytech Software, Inc., a Minnesota corporation. It was first introduced in early 2000 and was immediately a popular choice for computer monitoring needs. Years of listening to customer feedback and refinement has made SpyAgent into a world-class security solution that parents, families, schools, institutions, and corporations benefit from. SpyAgent has consistently proved to be a cutting-edge solution with its easy to use graphical user interface, innovative feature additions, and vigilant updates. pogil polar and nonpolar molecules answer key
Spytech SpyAgent will continue to be a leading computer monitoring solution for many more years to come. This is the most beautiful part of the key
Should you have any questions or troubles with SpyAgent, Spytech is here to help you. Our 24/7 helpdesk can solve any technical problem you are having, as well as schedule remote assistance so we can quickly connect to your computer and set things up for you and ensure everything is working properly. The key isn’t just saying "no"—it’s revealing a
This is the most beautiful part of the key. Water molecules, with their δ+ and δ- ends, would rather cling to each other through hydrogen bonds than invite greasy octane to the party. Octane molecules, equally happy, huddle together via London dispersion forces. The key isn’t just saying "no"—it’s revealing a hierarchy of forces: Hydrogen bonding > dipole-dipole > London dispersion. Water wins. Octane floats on top, living its best nonpolar life. The Ultimate "Answer Key" Insight If you look at a completed POGIL answer key, you’ll see boxes filled with "polar" and "nonpolar," EN differences, and "yes/no" for solubility. But the master key is actually a single sentence: A molecule is polar if it has polar bonds AND an asymmetric shape that prevents the dipoles from canceling. Everything else—miscibility, boiling point, surface tension—is just a consequence of that one rule. The answer key didn't give you facts; it gave you a lens. Now you can look at a molecule like chloroform (CHCl₃) and know: it’s polar, because while carbon is central, the three chlorines on one side and one hydrogen on the other break the symmetry.
Let’s walk through the "answer key" for a typical POGIL on polar and nonpolar molecules—but instead of just giving you the final column, let's see what the correct reasoning looks like. Question on the POGIL: Using electronegativity values, classify each bond as nonpolar covalent, polar covalent, or ionic: H-H, H-Cl, Na-Cl.
That’s the real answer key. It was never about the answers. It was about learning to see the invisible tug-of-war inside every bond.
Symmetry destroys polarity. Asymmetry creates it. This is why CO₂ is nonpolar (linear, symmetric) but SO₂ is polar (bent). The atoms themselves are less important than how they arrange themselves in space . Model 3: The Solubility Test (Like Dissolves Like) Question on the POGIL: Will octane (C₈H₁₈, nonpolar) dissolve in water?
You’ve just finished the POGIL activity. Your group argued about whether carbon dioxide is polar (it’s not, Carl, stop fighting it), you drew more partial charges (δ+ and δ-) than a thundercloud, and now you’re staring at the instructor’s answer key. You want the "right" answers. But here’s the secret the key doesn’t shout: The answers are just the destination. The models were the map.
Purchase SpyAgent and Start Monitoring Today! Risk-free Purchase - 15 day Money back Guarantee!
Download SpyAgent's installation software to your computer. Your download is available immediately after purchasing from our secure website.
Run SpyAgent's installer on the computer you want to monitor and customize your monitoring options to suit your needs.
Start monitoring your computer. View all recorded activities by accessing SpyAgent on the monitored computer, or remotely via our cloud website.
This is the most beautiful part of the key. Water molecules, with their δ+ and δ- ends, would rather cling to each other through hydrogen bonds than invite greasy octane to the party. Octane molecules, equally happy, huddle together via London dispersion forces. The key isn’t just saying "no"—it’s revealing a hierarchy of forces: Hydrogen bonding > dipole-dipole > London dispersion. Water wins. Octane floats on top, living its best nonpolar life. The Ultimate "Answer Key" Insight If you look at a completed POGIL answer key, you’ll see boxes filled with "polar" and "nonpolar," EN differences, and "yes/no" for solubility. But the master key is actually a single sentence: A molecule is polar if it has polar bonds AND an asymmetric shape that prevents the dipoles from canceling. Everything else—miscibility, boiling point, surface tension—is just a consequence of that one rule. The answer key didn't give you facts; it gave you a lens. Now you can look at a molecule like chloroform (CHCl₃) and know: it’s polar, because while carbon is central, the three chlorines on one side and one hydrogen on the other break the symmetry.
Let’s walk through the "answer key" for a typical POGIL on polar and nonpolar molecules—but instead of just giving you the final column, let's see what the correct reasoning looks like. Question on the POGIL: Using electronegativity values, classify each bond as nonpolar covalent, polar covalent, or ionic: H-H, H-Cl, Na-Cl.
That’s the real answer key. It was never about the answers. It was about learning to see the invisible tug-of-war inside every bond.
Symmetry destroys polarity. Asymmetry creates it. This is why CO₂ is nonpolar (linear, symmetric) but SO₂ is polar (bent). The atoms themselves are less important than how they arrange themselves in space . Model 3: The Solubility Test (Like Dissolves Like) Question on the POGIL: Will octane (C₈H₁₈, nonpolar) dissolve in water?
You’ve just finished the POGIL activity. Your group argued about whether carbon dioxide is polar (it’s not, Carl, stop fighting it), you drew more partial charges (δ+ and δ-) than a thundercloud, and now you’re staring at the instructor’s answer key. You want the "right" answers. But here’s the secret the key doesn’t shout: The answers are just the destination. The models were the map.